Behind BIO’s 105% Surge: AI-Driven Drug Discovery Emerges—but Real-World Market Approval Remains Distant
TechFlow Selected TechFlow Selected

Behind BIO’s 105% Surge: AI-Driven Drug Discovery Emerges—but Real-World Market Approval Remains Distant
The first FDA approval for AI-driven drug development will not occur until 2028.
Author: Muhammad Yusuf, Research @Delphi_Digital
Compiled by TechFlow
TechFlow Intro: The AI agent peptAI designed a novel ADHD candidate drug in 24 hours; BIO Protocol’s token surged 105%; and “DeSci” flooded crypto Twitter overnight. Yet stepping back to examine the data: While AI-discovered drugs do achieve an impressive 80–90% Phase I clinical trial success rate, Phase I only tests whether a drug kills you—not whether it cures anything. For the pivotal Phase III trials—the true determinant of therapeutic efficacy—not a single one among the world’s 173 AI drug pipelines has been completed. The most optimistic forecast sees the first FDA approval no earlier than 2028, while crypto markets can’t sustain attention for even six months. The endgame? Either DeSci develops financing mechanisms aligned with the four-year clinical development timeline—or another narrative bubble bursts.
AI-Driven Drug Discovery
An AI agent named peptAI designed a novel peptide-based ADHD candidate drug from scratch in 24 hours, ran it through eight validation steps, and output a molecule ready for wet-lab testing—costing only a few thousand dollars in lab work. BIO Protocol’s token surged 105%. Within hours, half of crypto Twitter added “DeSci” to their bios—just as they’d all added “AI” six months earlier.
Open-source protein-folding models now match AlphaFold3’s performance at zero licensing cost. Public bioactivity databases cover 2.5 million compounds. Wet-lab validation costs less than $2,000. AI is compressing both the cost and time of drug discovery.
I spent a week trying to understand what’s genuinely different this time.
Passing Phase I Is Not Impressive
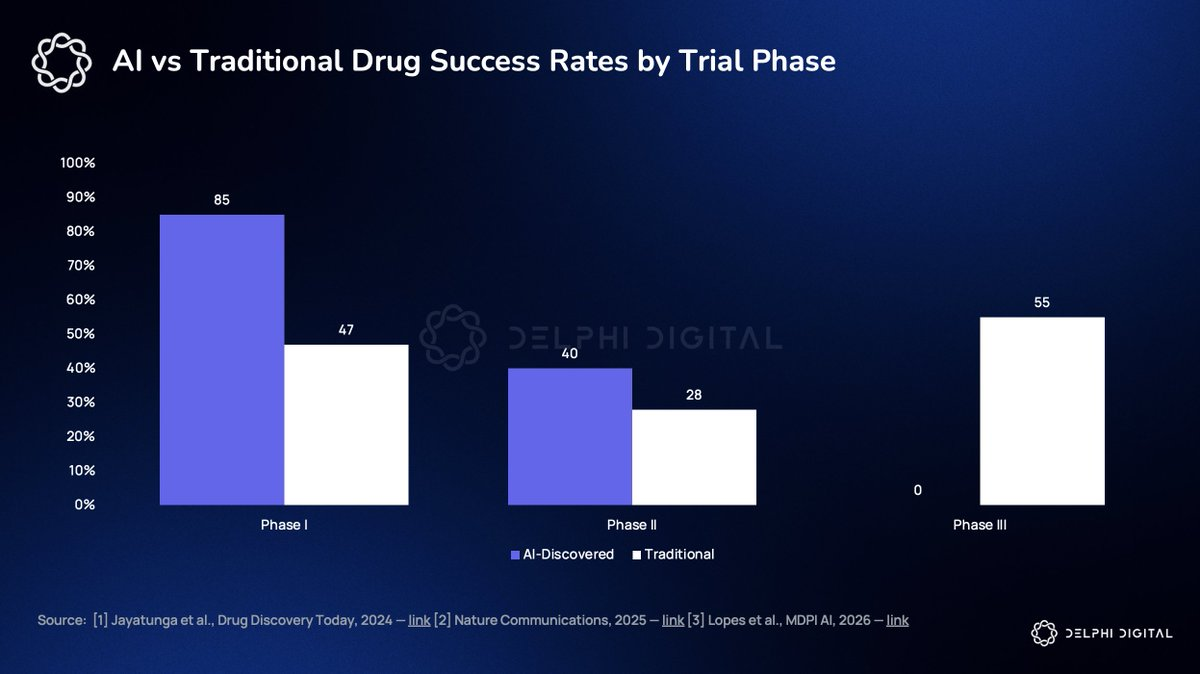
The oft-cited statistic—that AI-discovered drugs achieve 80–90% Phase I success versus ~47% for traditional drugs—is misleading without context. Phase I only assesses safety (“Will this kill you?”), not efficacy (“Will this cure you?”). Passing Phase I merely means your compound is safe enough to proceed—it still must navigate the entire remaining path to FDA approval.
Fewer than 40 AI-discovered compounds have reported Phase II data; none have completed Phase III. Insilico Medicine’s Rentosertib is currently the most advanced AI-discovered compound: It published positive Phase IIa results for idiopathic pulmonary fibrosis in Nature Medicine in mid-2025, and began Phase III enrollment in China in Q4 2025. Even under perfect conditions (enrollment completed in 2027, data readout in 2028, FDA review in 2029), that’s at least three years—and Rentosertib is the strongest candidate among the 173 AI drug pipelines. Several others were shelved in 2025 after failing endpoints in atopic dermatitis, schizophrenia, and oncology. Independent analysts estimate a 60% probability that the first AI-designed drug receives FDA approval by 2027—but none has yet completed the full regulatory pathway.
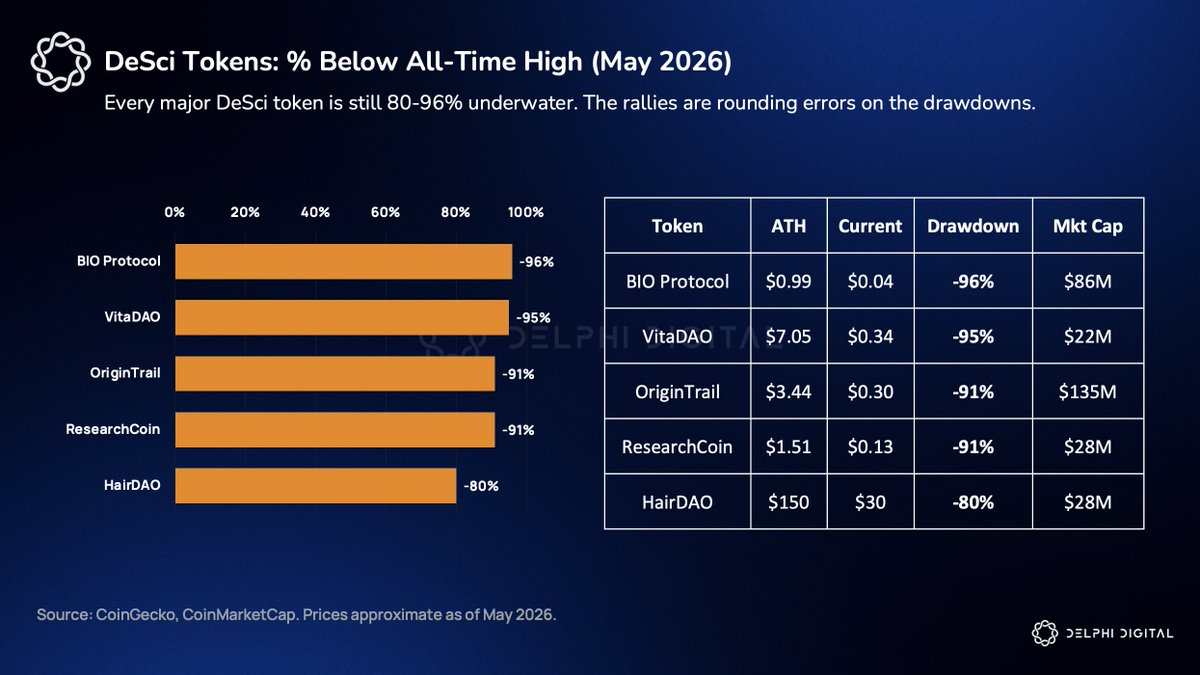
Can Crypto Twitter Sustain Real DeSci?
Keep those timelines in mind—and then look at BIO Protocol’s price chart. The token dropped from $0.89 to $0.018, then surged 105% on the peptAI announcement. $720 million in trading volume churned through a $68 million market cap. The entire DeSci funding thesis rests on token holders patiently waiting 7–10 years for clinical outcomes—while crypto Twitter abandons narratives long before Phase I data is unblinded.
Pump Science leaked its private key on GitHub, spawning numerous scam tokens—including one literally named “Cocaine.” The enforceability of IP-NFTs has never been tested in court.
Open Science vs. DeSci
If we avoid reflexive self-deception and chronic speculation, DeSci retains a sliver of hope within open science.
In October 2025, the OpenFold Consortium released OpenFold3 under the Apache 2.0 license—fully trainable, commercially usable, built on over 300,000 experimentally determined structures (unlike AlphaFold3, which Google restricts to academic use). MIT and Recursion jointly launched Boltz-2, predicting protein structure and binding affinity 1,000× faster than physics-based methods. The Baker Lab released RFdiffusion3 in December. ChEMBL hosts 2.5 million bioactive compounds with full ADMET profiles—freely accessible to anyone with a laptop. Infrastructure once costing pharmaceutical companies millions to build in-house now resides on GitHub under permissive licenses. Five pharma companies are now federating proprietary drug-protein libraries via the OpenFold3 Federation program. Crypto Twitter ignores these developments—there’s no token to trade. And I genuinely doubt core contributors to these codebases would get excited about tokens.
The Research Funding Crisis
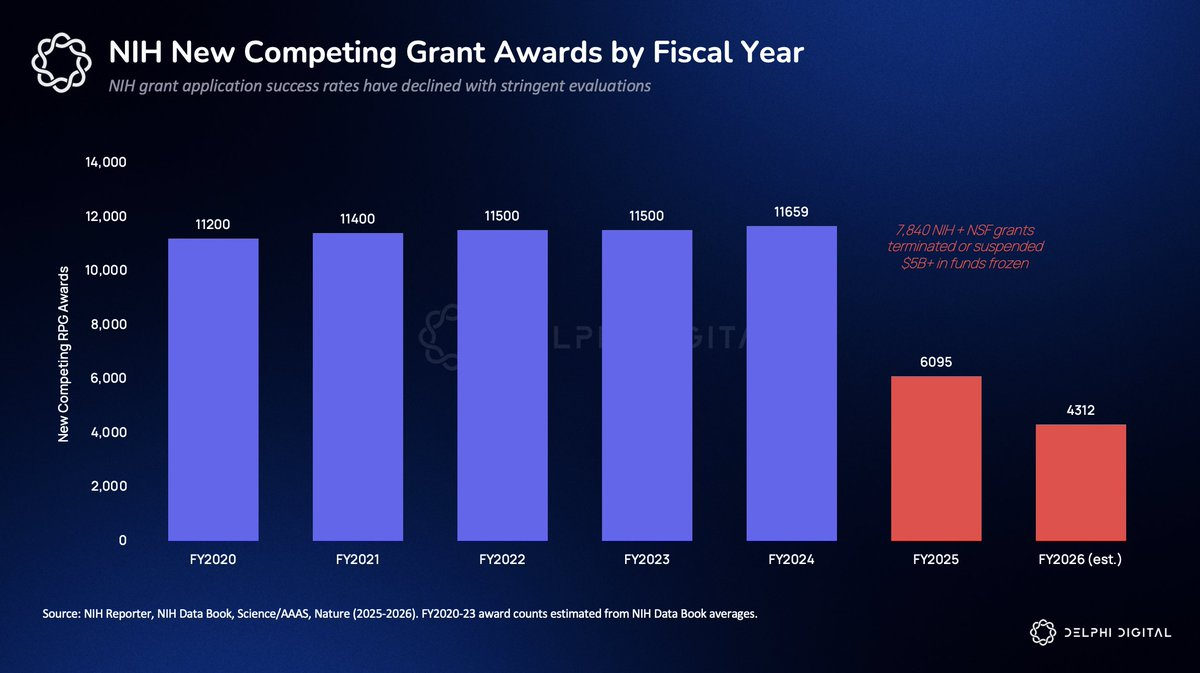
In 2025, over 7,800 NIH and NSF grants were terminated or suspended, freezing more than $5 billion in funding. The NIH—the world’s largest public funder of biomedical research (~$47 billion annually)—has flatlined its budget due to congressional appropriations, yet the government continues freezing active pipelines. New competitive grants plummeted from 11,659 in FY2024 to just 6,095 in FY2025—a 48% drop. Grant application success rates fell from 21% to 13%. Fred Hutch lost $508 million; Harvard lost $945 million.
This funding vacuum is why DeSci has an opening—if executed correctly. In July 2025, VitaDAO-funded Gero signed a research and licensing agreement with中外 Pharmaceutical (a Roche subsidiary, valued at ~$100 billion—not a meme), with milestone payments up to $250 million. This marks the first time a DAO-funded project produced something a major pharmaceutical company valued in the nine-figure range. The deal closed without governance disputes or rug pulls—and remains the most significant event yet in the field.
Four Years Is Eternity
This year, 15–20 AI-discovered drugs will enter Phase III. Rentosertib’s data won’t be available until 2028—meaning several more years will pass before we know whether any of this translates into real human efficacy. Regardless of token presence, open-source tooling will keep driving down costs; the funding vacuum will continue pushing researchers toward whoever writes checks. Open protein-folding models now match AlphaFold3 at zero licensing cost; wet-lab validation costs under $2,000; and the NIH just recorded its lowest grant success rate in two decades. Even if AI delivers on every promise—halving drug development timelines—you still face a 4–5-year discovery-to-approval window, assuming improved Phase III success rates. Four years is eternity in an industry where portfolio convictions rotate with quarterly earnings calls—and token holders treat a six-month holding period as life imprisonment.
Whether or not anyone buys tokens, the cost of discovery and innovation keeps falling each quarter. The NIH-induced funding vacuum is also real. Between these two forces lies a viable path: tokens funding specific clinical trials with defined milestones, governance entrusted to domain experts. The Gero/中外 Pharma deal proves DAO-funded projects can generate nine-figure commercial value. Setting aside hype—I wonder whether anyone will build defensible financing infrastructure for genuine DeSci.
Join TechFlow official community to stay tuned
Telegram:https://t.me/TechFlowDaily
X (Twitter):https://x.com/TechFlowPost
X (Twitter) EN:https://x.com/BlockFlow_News